Hertz cathode ray experiment6/21/2023 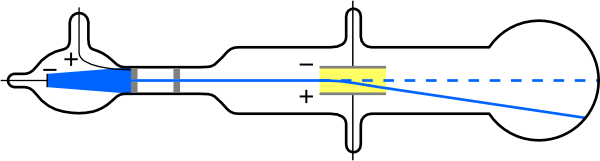
This cathode ray also became known as an ‘electron gun’. Physicists in the 19th century found out that if they constructed a glass tube with wires inserted in both ends, and pumped out as much of the air as they could, an electric charge passed across the tube from the wires would create a fluorescent glow. Look at any glowing neon sign or any ‘old-fashioned’ television set, and you are looking at the modern descendants of the cathode ray tube. X-rays are extensively used in medicine and industry to produce images of internal structures because they are absorbed by many forms of matter, including body tissues.Even without consciously realizing it, most of us are already aware of what a cathode ray tube is. The frequency of X-rays is higher than the frequency of ultraviolet light but less than that of a gamma ray. They have extremely short wavelength and high frequency with wavelengths ranging from about 10−8 to 10−12 meter and corresponding frequencies from about 1016 to 1020 hertz (Hz). Wilhelm Roentgen died on Februin Munich at the age of 77. This was a true acknowledgement of his remarkable discovery which was going to be highly beneficial for mankind in the coming years. In 1901, Roentgen received the first ever Nobel Prize in Physics. His discovery revolutionized the entire medical profession and set foundation for diagnostic radiology. Based on his subsequent research and experiments, he declared that X-ray beams are produced by the impact of cathode rays on material objects. He generated the very first "roentgenogram" by developing the image of his wife's hand and analyzed the variable transparency as showed by her bones, flesh and her wedding ring. He continued his experiments using photographic plate to capture the image of various objects of random thickness placed in the path of the rays. The screen became fluorescent even though it was placed in the path of the rays, two meters away from discharge tube. His experiment that involved working in a totally dark room with a well covered discharge tube resulted in the emission of rays which illuminated a barium platinocyanide covered screen. On Novemwhile he was experimenting, he observed that certain rays were emitted during the passing of the current through discharge tube. His experiments involved the passing of electric current through gases at extremely low pressure. 
Wilhelm Roentgen was already working on the effects of cathode rays during 1895, before he actually discovered X-rays. In later years, he was offered the Presidency of the Physikalisch-Technische Reichsanstalt at Berlin which he did not take and also refused to take the Chair of Physics of the Berlin Academy. In 1899, he also declined a similar offer from University of Leipzig as well. It is important to mention that he declined invitations for the chair of physics from the Universities of Jena and Utrecht in 18 respectively. His Professional Career YearĪcademy of Agriculture at Hohenheim, Wurttemberg In the same year, he assisted Kundt and followed him to Wurzburg and then to the University of Strasburg in 1873. He graduated from the University of Zurich and received his Ph.D. There he flourished greatly under the influence of the teachers like Kundt and Clausius. In 1865, he studied mechanical engineering at the Federal Polytechnic Institute in Zurich having failed to get admission in University of Utrecht lacking required credentials. There he got involved in a contrivance against one of his teachers and was expelled subsequently. In 1862, he joined Ambachtsschool a technical school in Utrecht. He was not a sparkling student rather he was keenly interested in nature during his young years. For his early education he went to a boarding school in Apeldoorn named, Institute of Martinus Herman van Doorn. He was raised in the Netherlands because his family moved to Apeldoorn when he was still three. Born on Main the small town of Lennep (Rhine Province) in Germany Wilhelm Conrad Roentgen was the only child of a cloth merchant.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
